synthetic routes for aromatic compounds
synthetic routes for aromatic compounds
The reaction shown in figure 4 below relate principally to aliphatic substance in which R represents either H or an alky group such as CH3. In many cases, R could also represent an aromatic group such as phenyl (C6H5). However, there are some reactions of aromatic chemicals (arenes) without parallel in aliphatic chemistry. In particular, these include the various substitutions of an aromatic ring. Thus, there is a separate range of synthetic routes of arenes. Some of the conversions linking the simpler aromatic chemicals such as benzene, methylbenzene (toluene) and phenol are shown in figure 4.1 below.
Drugs, detergents and dyes can all be made from benzene, which is unquestionably the most important arene. At one time, benzene, toluene and phenol were produced from coal tar. With the development of catalytic reforming, almost all benzene and toluene now come from oil.
There is one last point. In the industrial manufacture of chemicals, cost is all-important. Hence, cheap reagents such as water, hydrogen, sodium hydroxide and sulphuric acid are used in the production of ‘heavy’ organic chemicals whenever possible. Expensive reagents such as potassium dichromate(VI), lithium tetrhydridoaluminate(III)(LIALH4) and compounds are only used in small-scale laboratory preparations when the cost is relatively unimportant
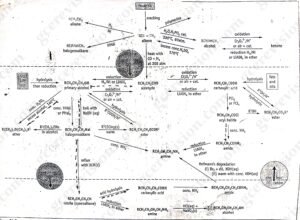
figure 4: important synthetic routes in organic chemistry
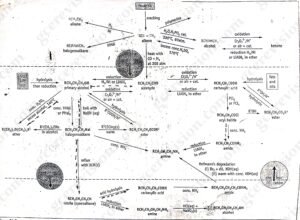
figure 4.1: important synthetic routes for arenes












Jay
December 3, 2021
Please when putting your logo on the papers try not to make them too bold cause I can't see everything that's being write on it